Crystal growth.
A crystal growth phenomena manifests itself in a long list of natural and technological processes.Crystals are solid materials with atoms located in regular position defined by long distance symmetry. The structure of crystals gives each of them special and often unique properties essential for material culture of civilization. The rate of transformation of atomic structure motive from chaotic in liquid, gas or other state into crystals with hard order structure plays important role in series of industrial productions of variety construction and other industrial materials as well for research of earth minerals formation in geology and in space bodies in cosmology. There is fast expanding industry for production of bulk single crystals for subsequent usage in electronics or for gems in jewelry.
Articles directly related to the understanding of crystal growth phenomena are presented here. The core of the content is outcome of from many years of Dr. Leonid Sakharov research and experience.
Content:
| A model of thermally activated reaction of crystal growth. | |
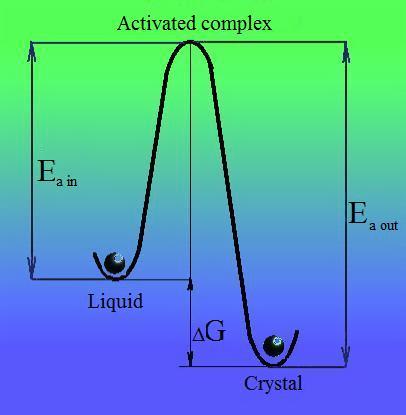 | The model of thermally activated crystal growth describes rate of crystal surface expansion under assumption model that all positions on it have equal chances to accept or emit a molecule. The model gives qualitatively adequate description for temperature dependence of growth rate versus temperature but failed to explain facet like growth near melting point and has to be considered as first approach in theory of crystal growth. |
| Thermodynamic potential of crystal growth. | |
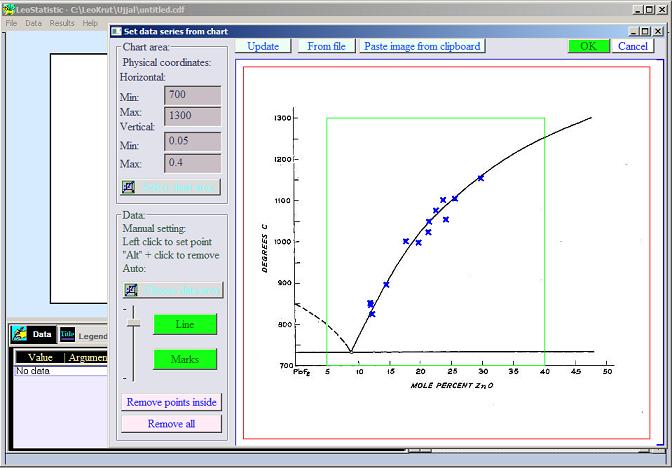 | The moving force of crystallization is difference between thermodynamic potentials of crystalline phase and its surrounding. There is direct connection expressed in formulas between amount of heat released during phase transformation (enthalpy), melting temperature and thermodynamic potential of crystal growth. The way for the defining enthalpy of crystal growth from phase diagrams are demonstrated on the example of ZnO. |
| Two-dimensional nuclei growth mechanism. | |
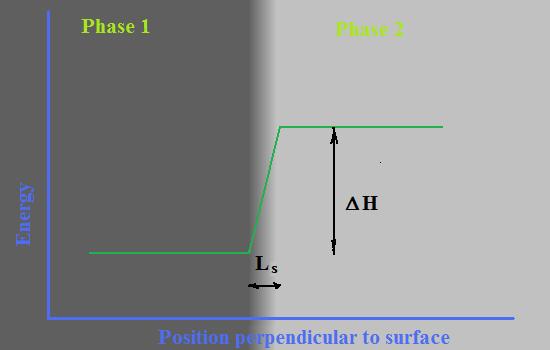
 | The model of two-dimensional nuclei spontaneously appeared on the surface of the crystal is the only visible explanation for layer by layer growth of the crystal that has its manifestation in smooth facets crystals. Formulas for the growth rate by two dimensional mechanism are derived from basic assumptions. |
| Estimation of crystal surface energy. | |
 | Surface energy of a crystal is in the same time one of the most important characteristics for any theoretical concept described a kinetic of crystal growth and is highly difficult for direct measurement in experiment. The simple formula for estimation of surface energy on the base of presumption that surface energy is concentrated inside depth of one atomic layer is presented and tested. |
| Phenomenology of crystal growth. | |
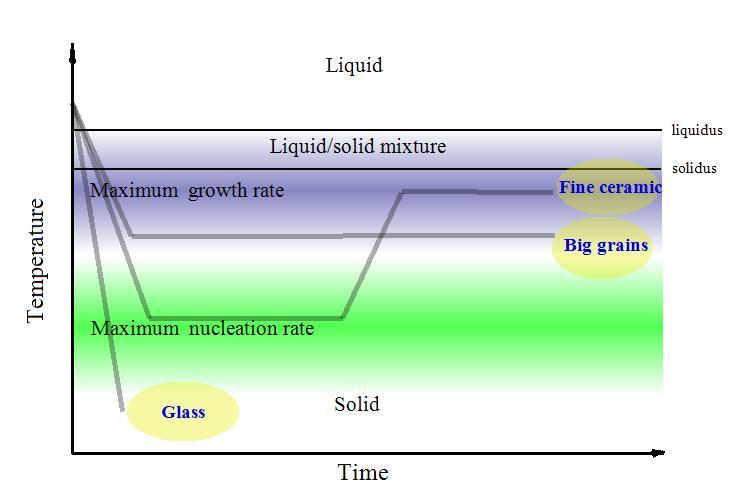 | The crystal growth has distinct features these can be described here without attracting theoretical concepts of mechanism of the phenomena. Wide variety for different crystals of rates for two its basic processes: nucleation – spontaneous appearance of small roots of crystals and their expansion explains great diversity of properties of solid matter in nature and industry. |
| Types of molecular structures on the crystal surface. | |
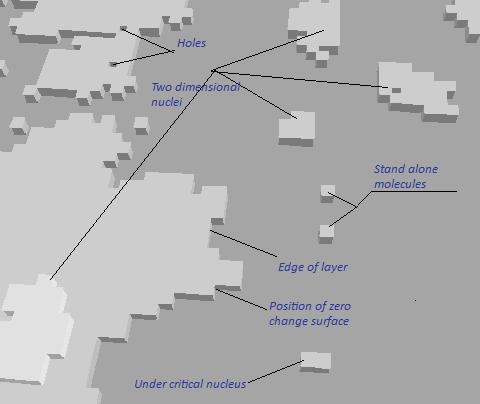 | A molecular structure of the crystal surface is defined the possible mechanisms of its growth. The rough surface produce continues mechanism, absolute smooth flat surface corresponds to two dimensional nucleation, a presence of dislocation leads to the spiral mechanism. |
Nucleation energy barrier at crystallization in nonstoichometric composition glass-forming solutions.
The dependence of the energy barrier of nucleation on the composition of the melt is considered. Under sufficiently general assumptions regarding the form of the dependence of the melt concentration in front of the crystal-melt boundary, taking into account the law of conservation of mass, a unique expression is obtained for the magnitude of the minimum change in the free energy due to the deviation of the melt composition from the initial one.
The dependence of the energy barrier of nucleation on the composition of the melt is considered. Under sufficiently general assumptions regarding the form of the dependence of the melt concentration in front of the crystal-melt boundary, taking into account the law of conservation of mass, a unique expression is obtained for the magnitude of the minimum change in the free energy due to the deviation of the melt composition from the initial one.
| Nucleation from own melt. | |
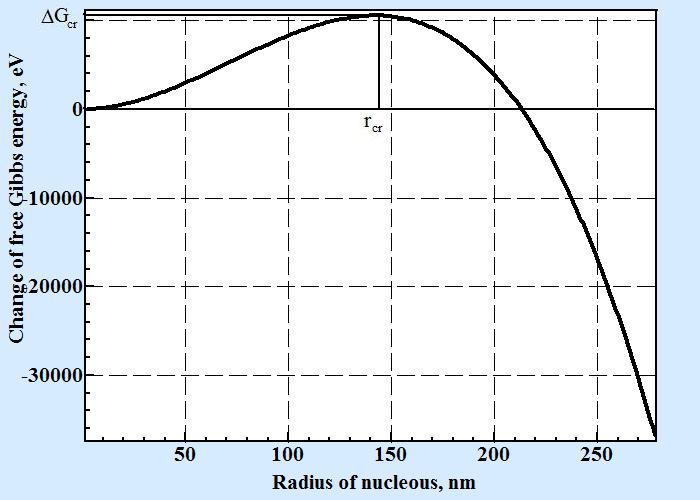 | Nucleation in melts happens in spite that thermodynamics for the process of spontaneous formation of crystal areas is prohibited due to influence of surface energy. Formulas for critical size nucleus and frequency of formation as fluctuation over critical nucleus are derived. These formulas can explain great variety observed in nature and industry rates of melts crystallization. |
| Dislocation induced crystal growth | |
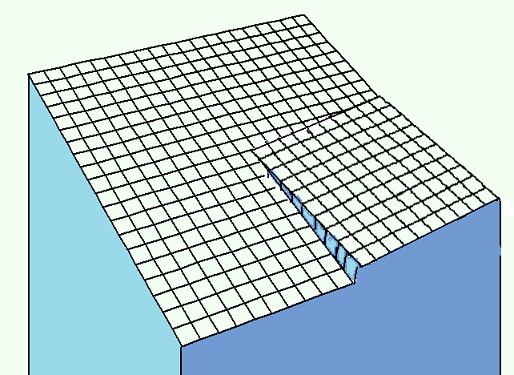 | The dislocation induced mechanism of crystal growth is based on premise that imperfections on the flat surface of the crystal can play the role of two dimensional nucleus bypassing necessity of their spontaneous appearance to produce layer by layer growth. For the special case of spiral dislocation a derivation of the analytical formula for the growth rate with square dependence from supercooling is presented. |